 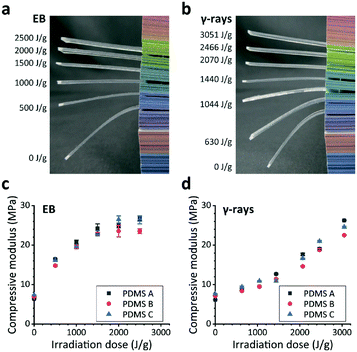 
While the results are irrefutable, the mechanism of how an electrically conducting scaffold interacts with an electroactive tissue remains has remained elusive. A growing trend in the use of conductive scaffolds for the growth of these tissues has been witnessed recently. Due to the high prevalence of cardiac and brain‐related ailments that involve some necrosis of tissue, cardiac and neuronal tissue engineering are intensely studied fields in regenerative medicine. Tissue engineering is a promising therapeutic approach in medicine, targeting the replacement of a diseased tissue with a healthy one grown within an artificial scaffold. Therefore, MCNTs might be more effective materials for accelerating bone formation even than nHA. Moreover, the MCNTs could induce ectopic bone formation in vivo while the nHA could not, which might be because MCNTs could stimulate inducible cells in tissues to form inductive bone better than nHA by concentrating more proteins including specific bone-inducing ones secreted from M2 macrophages. Most importantly, the MCNTs could induce osteogenic differentiation of the HASCs better than the nHA, the possible mechanism of which was found to be that the MCNTs could activate Notch involved signaling pathways by concentrating more proteins, including specific bone-inducing ones. In vitro culture of human adipose-derived mesenchymal stem cells (HASCs) on the MCNTs and nHA demonstrated that although there was no significant difference in the cell adhesion amount between on the MCNTs and nHA, the cell attachment strength and proliferation on the MCNTs were better. We hereby comparatively studied the osteogenic ability of our treated multi-walled carbon nanotubes (MCNTs) and the main inorganic mineral component of natural bone, nano-hydroxyapatite (nHA) in the same system, and tried to tell the related mechanism. But efficacy degree of some nanomaterials to promote specific tissue formation is still not clear. The biomimetic features and unique physiochemical properties of nanomaterials play important roles in stimulating cellular functions and guiding tissue regeneration. It has been well recognized that the development and use of artificial materials with high osteogenic ability is one of the most promising means to replace bone grafting that has exhibited various negative effects. Although MCNTs have not been examined as bone repair materials for a long time, there have been already a lot of related original publications. Following the discovery of multi-walled carbon nanotubes (MCNTs), one of the most representative nanomaterial, with unique electrical, mechanical, and surface properties, carbon-based nanotechnology has been rapidly developing as a platform technology for a variety of uses including biomedical applications, and up to now MCNTs appear well suited as biomaterials to enhance the properties and function of the medical devices, for example for improving tracking of cells, sensing of microenvironments, delivering of transfection agents and providing nanostructured surfaces for optimal integration with the host body, which is not only because of their ability to simulate dimensions of proteins that comprise native tissue using their unique properties but also because of their higher reactivity for cell interactions to improve the cellular functions. e) Histogram showing the similar average values for pore diameters and throat diameters for both samples (n = 3 for PDMS, n = 3 for PDMS+CNT). d) Histograms revealing that the two scaffolds have similar levels of porosity and isotropy. Both samples show that all empty spaces inside the material are interconnected. Right, the scaffold's matrices (in white) are shown. In blue and green, the maximal filling sphere for pore and throat diameter determinations, respectively, and in red, the interconnection path. c) Left, an example of ♜T volumetric reconstructions of scaffolds' pores and channelization in a ROI of 5123 voxels of the total volume. b) SEM image of a PDMS+CNT scaffold slice showing a similar porous morphology a high‐resolution image of the pore surface (top‐left inset), nicely illustrates the carpet of MWCNTs decorating exclusively pores' walls and resulting in a rough surface. The high‐resolution image of the pore surface shown in the top‐left inset highlights the PDMS smoothness. a) SEM image of a PDMS scaffold slice showing the typical “sponge‐like” aspect. PDMS and PDMS+CNT scaffolds: SEM and ♜T measures. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
